Three names in the vast and varying world of metals stand out amongst the crowd thanks to their unique reddish hues. These metals are known as the "red metals" and make up some of the most commonly used metals in the world: copper, brass, and bronze.
From construction and architecture to telecommunications and machinery, there is no denying the importance red metals have in our daily lives. Yet, for being some of the most important metals in existence, knowledge about red metals is pretty rare.
So, exactly what are red metals? And why are they so important?
What are Red Metals?
Though the red metals are all quite different in composition, quality, and application, they all have one unifying component: copper.
As one of the first metals mined by our ancestors, copper has been integral to the development of societies and serves as the base metal for the other two red metals: brass and bronze.

Copper is unique in that it is one of the few metals that can be used directly in its natural state. It's a soft metal known for its electrical and thermal conductivity, formability, and resistance to corrosion. Today it is most commonly found in electrical materials, roofing, plumbing, and industrial machinery.
In addition to its various applications, copper is used to creating red metal's brass and bronze. As red metal alloys, brass and bronze both consist of mixing copper with other alloying elements.
 Brass, for example, is created when adding varying amounts of zinc to copper. Depending on the zinc content, different qualities of brass can be created and used for different applications. The more zinc that is added to the mixture, the stronger, more ductile, and lighter in color the metal becomes. Brass is commonly used in architecture for its decorative features as well as in manufacturing, construction, electrical, and plumbing industries.
Brass, for example, is created when adding varying amounts of zinc to copper. Depending on the zinc content, different qualities of brass can be created and used for different applications. The more zinc that is added to the mixture, the stronger, more ductile, and lighter in color the metal becomes. Brass is commonly used in architecture for its decorative features as well as in manufacturing, construction, electrical, and plumbing industries.
Bronze, similar to Brass, is an alloy of copper, but instead of zinc, copper is combined with the tin and other alloying elements such as aluminum, silicon, manganese, and phosphorus. Of the different types of bronze, bearing bronze is one of the most popular, offering high corrosion resistance and hardness, making it ideal for industrial applications and hardware such as bearings and bushings.

How are Red Metals Made?
As one of the first metals to be discovered by our ancestors, copper has been used by humans for over 10,000 years. But over time, the way we use, process, and alloy copper has significantly changed.
Today, about 80% of copper is extracted from sulfide ores, where the copper is chemically bound with sulfur. In order to extract the copper from the ore, it must be refined through several different manufacturing processes. These processes involve the following:
- mining
- concentrating
- smelting
- refining
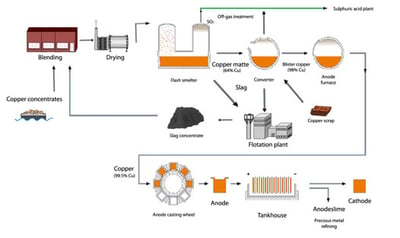
Once copper has been refined, it is cast into ingots, cakes, billets, or rods depending on its final application or can be used to produce a copper alloy (most commonly, brass or bronze).
As copper alloys, brass and bronze both contain their own unique manufacturing processes that depend upon the desired shape and properties of the metals. The typical production of both requires heating and cooling processes where copper and specific amounts of alloying elements are melted, mixed, and cast for the final application.
Common Types of Red Metals
1. High Conductivity Copper: known as pure copper, and must contain a minimum of 99.3% Cu. This type of copper is generally used for electrical applications such as wire and cable because of its excellent ductility and high electrical conductivity.
There are a number of different grades of HC copper that differ slightly in purity, but the most common grade is known as ETP copper (electrolytic tough pitch), UNS C11000. This copper contains a minimum of 99.90% Cu.
2. Deoxidized Copper: as the name suggests, the oxygen in this type of copper is removed. This gives it the ability to be brazed or welded without fear of embrittlement. Deoxidized copper is generally found in building construction, used in central heating systems, tubes for gas and water supply, and sheeting for roofs. When weathered, these coppers form what is known as patina (green or brown film produced by prolonged weathering) and are often admired for their aesthetic effects.
3. Copper Alloys: combining elements with copper allows for a broad range of different combinations that each have their own unique set of properties and applications. The most common alloying elements used include zinc, tin, nickel, and aluminum and create the following types of metal alloys:
- Brass: copper + zinc
- Red Brass (Gunmetal): copper + tin + zinc
- Bronze: copper + tin
- Phosphor Bronze: copper + tin + phosphorus
- Aluminum Bronze: copper + aluminum
- Copper-Nickel: copper + nickel
- Nickel Silver: copper + nickel + zinc
Why use Red Metals?
By now, you should have a solid idea of what red metals are and how they are made, but here comes the big question: why do we continue to use them?
Like aluminum, copper is recyclable without any loss of quality, both from its raw state and from manufactured products. In fact, an estimated 80% of all copper ever mined is still in use today!
In addition, red metals offer a wide spectrum of qualities for practical application across multiple industries. Combining their sustainability with this type of versatility makes red metals some of the most important metals in the world. Red metals are an important part of sheet metal fabrication, and they are often used in projects that require strength and durability. As a metal company, we offer a wide variety of red metals, including copper, brass, and bronze, which can be seen on our metal chart.
From construction and architecture to telecommunications and machinery, these metals contribute to both the development of our society and the betterment of our lives.
If it turns out that this wasn't everything you wanted to know and more, check out our FREE Digital Stock Book for all your processing needs by clicking on the image below.


Our indexed and searchable PDF makes it easy to quickly locate the information you need.
What's Inside?
- Specifications for industry-standard products
- Common conversion charts and guides
- Available processing services by product type
Image Sources:
1 https://bulgaria.aurubis.com/our-business/



