Aluminum is the most widely used nonferrous metal in the world. Its production and application exceed all other metals except iron and steel—and for good reason. It is lightweight, corrosion-resistant, and thermally and electrically conductive and available in a wide range of alloys engineered for specific performance requirements.
This guide covers what aluminum is, how it is made, which alloy series are used for which applications, what Boyd Metals stocks, and how to determine whether aluminum is the right material for your project.
What is Aluminum?

Aluminum in its pure form is soft, ductile, and highly reactive with oxygen. When exposed to air, pure aluminum immediately forms a thin oxide layer—a property known as aluminum reactivity—that actually contributes to its natural corrosion resistance in most environments. This oxide layer is stable and self-renewing, which is why aluminum performs well in outdoor and aquatic applications without additional coating.

Pure aluminum is not found free in nature. It exists as aluminum oxide compounds within bauxite ore, a rocky material mined from the Earth's crust. Once extracted, pure aluminum is almost always combined with other alloying elements such as copper, magnesium, manganese, silicon, tin, or zinc—to improve strength, hardness, weldability, or other properties required for commercial use.

The aluminum products you encounter in industrial applications—sheet, plate, structural shapes, tubing—are aluminum alloys, typically composed of 90–99% aluminum with the remainder made up of those alloying elements.
How Is Aluminum Made?
Aluminum cannot be extracted from ore through simple smelting the way iron can. Two processes, developed in the 1880s, form the backbone of modern aluminum production.
Bayer Process
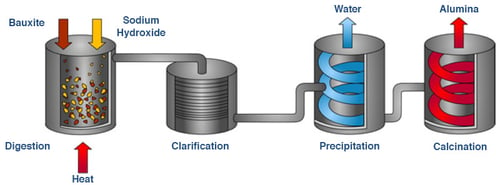
Raw bauxite is mined, crushed, blended, and converted into a slurry. That slurry is treated with heat and pressure to separate aluminum oxide (alumina) from water and impurities. What remains is a refined alumina powder ready for smelting.
Hall–Héroult Process
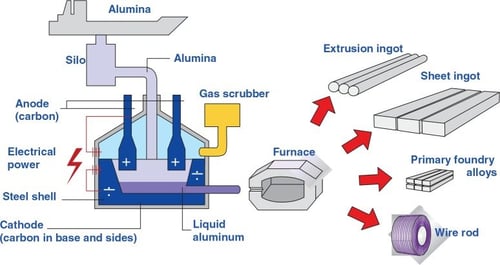
The alumina from the Bayer Process is dissolved in a molten mixture and electrolyzed at high energy. This causes aluminum and oxygen atoms to separate, producing metallic aluminum. The raw aluminum is then cast into billets or ingots for alloying and further processing.
Why Recycling Matters
Primary aluminum production is energy-intensive. Recycling aluminum requires only about 5% of the energy needed to produce it from ore, making it one of the most economically and environmentally efficient metals to recover and reuse. Aluminum is indefinitely recyclable without meaningful degradation in quality.
Aluminum Alloy Series: What the Numbers Mean
Aluminum alloys are classified by a four-digit numbering system. The first digit identifies the primary alloying element and the general alloy series. Understanding the series helps narrow down which alloy is appropriate for a given application before going further into specific grades and tempers.
Alloys fall into two broad categories: heat treatable (strengthened through controlled heating and quenching) and non-heat treatable (strengthened through cold working during rolling or forming).
-
Commercially Pure (Non-Heat Treatable): Aluminum at 99% or greater purity. Excellent corrosion resistance, high thermal and electrical conductivity, and good workability. Used primarily in electrical transmission lines and chemical processing equipment, where conductivity and corrosion resistance matter more than strength.
-
Copper Alloys (Heat Treatable): Copper is the primary alloying element. These alloys offer high strength and good toughness but reduced corrosion resistance compared to other series. Commonly specified in aerospace and aircraft structural components.
-
Manganese Alloys (Non-Heat Treatable): Manganese is the primary alloying element, often with small amounts of magnesium. Moderate strength, good workability, and good corrosion resistance. Common in beverage cans, HVAC ducting, and cooking equipment.

-
Silicon Alloys (Non-Heat Treatable): Silicon lowers the melting point of aluminum without creating brittleness. Used primarily in welding wire and filler materials and in some structural applications.
-
Magnesium Alloys (Non-Heat Treatable): Magnesium is the primary alloying element. These alloys offer moderate to high strength, excellent weldability, and strong corrosion resistance—particularly in marine and aquatic environments. Common in boat hulls, pressure vessels, and building construction.
-
Silicon + Magnesium Alloys (Heat Treatable): The most widely used series for structural and architectural applications. 6061 and 6063 are the dominant grades. Versatile, weldable, formable, and corrosion-resistant. Used in structural framing, transportation components, and general fabrication.
- Zinc Alloys (Heat Treatable): Zinc is the primary alloying element, with additions of magnesium, copper, or chromium. These alloys achieve the highest strength levels of any aluminum series and are used in commercial aviation and high-performance structural applications where the weight-to-strength ratio is critical.
Aluminum Formats Boyd Stocks
Boyd Metals stocks aluminum in the following formats and series. All availability is subject to location.
Structural Shapes
- Beams: I-beams and wide flanges in 6061-T6, 25′ lengths
- Angles: 6000 series in 16′ (6063) and 25′ (6061) lengths
- Channels: 6000 series in 16′ (6063) and 25′ (6061) lengths
- Tees: 6000 series in 16′ (6063) and 25′ (6061) lengths
Bar Stock
- Flats, rounds, squares, and hexes in 6000 series, 12′ and 16′ (6063) lengths
Plate
- 3000, 5000, and 6000 series in mill finish, various widths and lengths
Floor Plate
- 3000 and 6000 series in bright and mill finish, various widths and lengths
Sheet
- 3000, 5000, and 6000 series in mill finish, various widths and lengths
Tubing
- Round, square, and rectangular in 6000 series: 6061 in 24′ lengths, 6063 in 21′-1″ lengths
When Aluminum Is the Right Choice

Several factors influence aluminum pricing and lead time on any given order:
Alloy and temper. Common grades like 6061-T6 are widely stocked and typically available off the shelf. Less common grades, tempers, or sizes may require mill orders with longer lead times.
Form factor. Sheets and plates are generally the most readily available forms. Structural extrusions (angles, channels, tees) in less common sizes may require lead time.
Market conditions. Aluminum pricing is tied to LME (London Metal Exchange) spot prices and energy costs, since smelting is energy-intensive. Price volatility can affect both primary and recycled aluminum costs.
Order quantity. Standard mill lengths and quantities are the most cost-efficient. Custom lengths or tight size tolerances may increase cost and lead time.
How Boyd Metals Processes Aluminum
Boyd Metals offers several in-house processing services applicable to aluminum. Processing can be discussed as part of your material order to reduce outside fabrication steps.
Plasma Plate Cutting: Boyd's plasma cutting equipment handles aluminum plates. CNC-controlled plasma provides consistent tolerances, reduced slag, and faster turnaround than oxyfuel processes.
High-definition plasma can achieve tolerances of +/- .0625″ on aluminum and stainless steel in addition to carbon steel.
Shearing: Boyd's shears handle material up to 1/4″ thick and 144″ long. Both manually and CNC-controlled shears are available for aluminum sheets and light plates.
Sawing: CNC programmable saws with a capacity of up to 24″ square are used for aluminum bars, structural shapes, and tubing. Miter cutting capability is available.
Laser Cutting (2D and 3D): Boyd's laser cutting services are capable of processing aluminum sheet and plate.
Contact your nearest Boyd Metals location to discuss material availability and applicable processing services for your project.
Next Steps
Boyd Metals stocks aluminum sheet, plate, structural shapes, bar, and tubing in 3000, 5000, and 6000 series alloys. If you are sourcing aluminum sheet metal, structural aluminum, or custom-cut aluminum components, contact us to discuss your specifications, quantities, and lead time requirements.
Image Sources:
1 http://muharraq27.blogspot.com/2010/12/aluminium-processing.html
2 https://recyclenation.com/2014/03/recycle-aluminum/
3 https://www.lightmetalage.com/news/industry-news/recycling-remelt/hydro-starts-new-recycling-line/
4 https://www.indiamart.com/cmeri-durgapur-durgapur/






